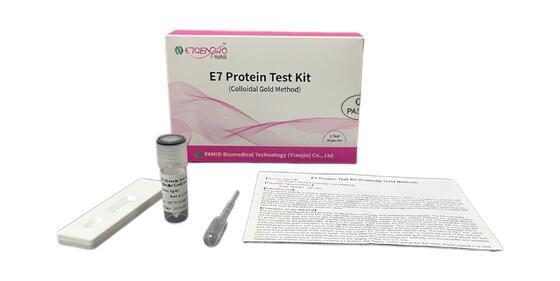
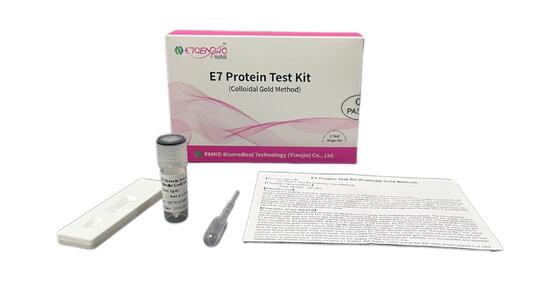
Zostaw wiadomość
Oddzwonimy wkrótce!
 Twoja wiadomość musi mieć od 20 do 3000 znaków!
Twoja wiadomość musi mieć od 20 do 3000 znaków!
 Proszę sprawdzić email!
Proszę sprawdzić email!
Zatwierdź
Więcej informacji ułatwia lepszą komunikację.
Pan
- Pan
- Pani
dobrze
Przesłano pomyślnie!
Oddzwonimy wkrótce!
dobrze
Zostaw wiadomość
Oddzwonimy wkrótce!
 Twoja wiadomość musi mieć od 20 do 3000 znaków!
Twoja wiadomość musi mieć od 20 do 3000 znaków!
 Proszę sprawdzić email!
Proszę sprawdzić email!
Zatwierdź